Solanum acaule

| Common Names | Apharu papa, Apharuma, Arakk papa, Atokk papa, Aya papa, Papa de Perdiz, Jupaypa papan, Khapharu ch’ogue, Machocc papa, Shiri, Shoco papa, Papa de las nubes |
| Code | acl |
| Synonyms | S. depexum, S. punae, S. schreiteri, S. uyunense |
| Clade | Mixed |
| Series | Acaulia |
| Ploidy | Tetraploid (4x) |
| EBN | 2 |
| Tuberization Photoperiod | Short Day |
| Self-compatibility | Yes |
| Nuclear Genome | A |
| Cytoplasmic Genome | M |
| Citation | Bitter: Repert. Spec. Nov. Regni Veg. 11: 391. 1912. |
Description
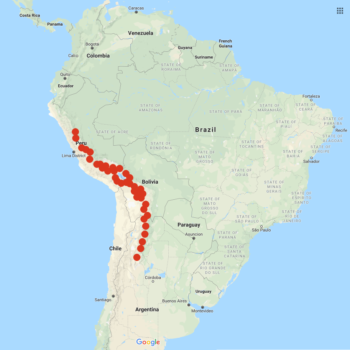
Solanum acaule is a wild potato species that ranges from central Peru through Bolivia and into northern Argentina. There may also be a population in Ecuador although the identity of this population is contested. This is the greatest natural distribution of any of the wild potato species (Ochoa 1990). This species grows largely at high altitudes, ranging from about 7900 to 15400 feet (2400 to 4700 m). A large part of its natural range is in the Puna, a highland region of Peru and Bolivia where the annual average temperature is only about 45 F (7 C). In the wild, it acts as a ground-cover plant, growing in large mats, and survives at higher elevations than any other potato species (Ochoa 2004).
The specific epithet, acaule, means “without stem,” referring to the generally low growing habit of this species. It is formed from the Latin prefix “a,” for “without,” and “caulis,” for “stem.” While there is no completely standardized pronunciation for scientific names, the most common way to pronounce this species is probably so-LAY-num ah-KAW-lay.
The growth habit of S. acaule can change significantly depending on climate (Ochoa 2004). The plants have a low growing habit, usually not exceeding eight inches in height in cold, high elevation conditions. They may form much longer and taller stems in warmer climates. Plants occasionally have no noticeable aerial stem at all, just a flat rosette growing against the ground. Flower color ranges from blue to violet or is occasionally white. Unlike most potatoes, S. acaule lacks a floral abscission zone (or flower joint). While this is not a trait of much importance to the breeder, it may be helpful in identifying mixed progeny. Stolons are typically two to three feet long. Tubers are typically round, white, and 1/2 to 1 inch in diameter.
S. acaule is most likely an allotetraploid (more specifically, an amphidiploid) as it has disomic inheritance. This means that the chromosomes segregate in pairs, dividing two ways as if the plant were a diploid. This is an indication that it probably originated as a hybrid of two different diploid species and has retained their independently segregating genomes. This probably explains why it is self-compatible when most potato species are not.
S. acaule has historically been assigned a number of subspecies, including subsp. acaule, subsp. aemulans, subsp. albicans, and subsp. punae. Some of these are no longer recognized or have been raised to species. Most populations of this species are tetraploid (4x). Hexaploid (6x) populations of S. acaule have been reported, but there is some doubt about whether they are actually S. albicans, a similar and closely related species that is normally hexaploid.
It is often noted that S. acaule bears both morphological and genetic similarities to S. demissum, which is a North American species that appears to have arisen from a similar evolutionary process. They are both allotetraploids, although from a different combination of species.
Subramanian (2017) found that at least some accessions of this species have unusually high dry matter content.
The S. acaule chloroplast genome has been sequenced and has a size of 155,570 bp, encoding 158 genes of which 105 code proteins, 45 tRNA, and 8 rRNA (Park 2021).
Resistances
S. acaule can have impressive frost tolerance; resistance to viruses PVX, PVY, PLRV, and PSTV; and potato cyst and root knot nematode resistance. A resistance gene for Potato Virus X, Rx2, has been identified in this species.
This species can resist frosts down to 22 degrees F (-5.5 C) (Sukumaran 1972) or perhaps even down to 20 degrees (-6.5 C) (Li 1977). Vega (1995) found that 100% of 351 accessions of this species survived through two light frosts with no damage. The rosette-like growing habit of this species keeps the plant close to the ground and probably aids in tolerance to low temperatures, but this is not a sufficient explanation. Chen (1977) found three structural differences between S. acaule and S. tuberosum that may account for its frost resistance. It has significantly thicker cell walls than S. tuberosum. Its chloroplasts have a much larger number of osmiophylic globuli and these globuli increase in number following frost exposure. The chloroplasts also become depleted of starch following frost exposure. This suggests the conversion of starch to soluble sugars, a common adaption to reduce the freezing point of plant cells. Taken together, these features suggest that S. acaule may be able to more effectively delay, survive, and recover from frost induced dehydration.
| Condition | Type | Level of Resistance | Source |
|---|---|---|---|
| Alternaria solani (Early Blight) | Fungus | Resistant | Prasad 1980 |
| Alternaria solani (Early Blight) | Fungus | Somewhat resistant | Jansky 2008 |
| Clavibacter michiganensis (Ring Rot) | Bacteria | Somewhat resistant | Laurilla 2003 |
| Drought | Abiotic | Somewhat resistant | Machida-Hirano 2015 |
| Frost | Abiotic | 21 to 23 F | Chen 1977 |
| Globodera pallida (Pale Cyst Nematode) | Invertebrate | Somewhat resistant | Jackson 1988, Bachmann-Pfabe 2019, Castelli 2003, Machida-Hirano 2015 |
| Globodera rostochiensis (Potato Cyst/Golden Nematode) | Invertebrate | Somewhat resistant | Castelli 2003 |
| Heat | Abiotic | Somewhat resistant | Machida-Hirano 2015 |
| Leptinotarsa decemlineata (Colorado Potato Beetle) | Invertebrate | Somewhat resistant | Machida-Hirano 2015 |
| Meloidogyne incognita (Root Knot Nematode) | Invertebrate | Resistant | Nirula 1967 |
| Meloidogyne spp. (Root Knot Nematode) | Invertebrate | Somewhat resistant | Machida-Hirano 2015 |
| Pectobacterium carotovorum (Blackleg/Soft Rot) | Bacteria | Some resistance | Chung 2011 |
| Phytophthora infestans (Late Blight) | Fungus | Somewhat resistant | Bachmann-Pfabe 2019 |
| Potato Leaf Roll Virus (PLRV) | Virus | Resistant | Machida-Hirano 2015 |
| Potato Spindle Tuber Viroid (PSTVd) | Virus | Somewhat resistant | Machida-Hirano 2015 |
| Potato Virus X (PVX) | Virus | Resistant | Machida-Hirano 2015, Wetter 1971 |
| Potato Virus Y (PVY) | Virus | Somewhat resistant | Machida-Hirano 2015 |
| Potato Virus Y (PVY) | Virus | Not resistant | Cai 2011 |
| Ralstonia solanacearum (Bacterial Wilt) | Bacteria | Somewhat resistant | Machida-Hirano 2015 |
| Synchytrium endobioticum (Wart) | Fungus | Resistant | Machida-Hirano 2015 |
Glykoalkaloid content
Osman (1986) determined that the primary glycoalkaloids in Solanum acaule are demmissine and tomatine and total glycoalkaloids ranged from 35 to 126mg/kg (Osman 1978). Van Gelder (1988) agreed that demissine and tomatine were the primary glycoalkaloids, along with a smaller amount of solanidine in one accession and TGA ranged from 12 to 85 mg / 100 g. Some varieties fall below the 20 mg / 100 g safety limit and this agrees with my taste buds.
I have tasted tubers from many different plants of different accessions. Few of these were bitter. Many of them have a sort of sour aftertaste at the back of the tongue and throat. I would guess that this is the flavor of tomatine. I have made several selections of this species that are palatable and taste indistinguishable from domesticated varieties, lacking any aftertaste.
Images
Cultivation
I have often found seeds of Solanum acaule slow and difficult to germinate, taking two to three times as long as seeds of domesticated varieties. Germination may require treatment with gibberellic acid (GA3). The US Potato Genebank found that some accessions had no germination without a 24 hour soak in 2000PPM GA3 (Bamberg 1999). Even with GA3 treatment, many seeds seem slow to germinate. S. acaule might require different conditions than are generally used for S. tuberosum.
Bamberg (1995) found that at least some accessions of S. acaule are able to produce a large amount of viable pollen even when temperatures exceed 100 degrees F for several hours during the day. However, flowering was reduced to a very low level under these conditions.
Many varieties produce berries at ground level, even becoming partly covered in soil (Ochoa 2004). This can be a disadvantage when growing at lower elevation where the berries may be eaten, particularly by slugs. Because this species lacks a flower joint, the berries are more difficult to harvest. You might need a knife or scissors to collect berries that ripen before the plant dies back.
Chen (1976) found that this species reaches its maximum frost resistance (about 15 degrees F, -9.3 C) following a three week period of shortening photoperiod and reducing day and night temperatures.
Bamberg (2017) found an 80% increase in seed set in this species with supplemental applications of liquid fertilizer at four and seven weeks after potting.
Towill (1983) summarized seed germination data from the USDA Potato Introduction Station and found that S. acaule retains excellent germinability over long storage, with almost no reduction after periods as long as 27 years at 1 to 3 C (about 34 to 37 F).
Tuber dormancy of five to six months is common in this species. Tubers will remain dormant for at least 16 months when stored at near-freezing temperatures.
Breeding
Frost resistance is the primary interest of modern potato breeders in Solanum acaule. The plant grows in very challenging environments where frosts can occur throughout the growing season and drought is also frequently a problem.
Ochoa (2004) describes S. acaule as being essentially cleistogamous (exclusively self-pollinating) as a consequence of its self-compatibility, short style, and early anthesis. Because of this, it should be emasculated when it will be used as a female parent in crosses.
Crosses with S. tuberosum
S. acaule has been used extensively in potato breeding. Varieties that include S. acaule in their pedigrees include Alaska Frostless, Bounty, Chieftain, Conestoga, Dakchip, Islander, Norchip, Nordak, Norgleam, Norking Russet, Redsen, Sangre, Snowflake, Trent, Viking, Yukon Gold (Plaisted 1989), Century Russet, Chieftain, NorDonna, and NorValley (Love 1999).
Alaska Frostless is probably the most frost resistant hybrid between S. acaule and S. tuberosum. Unfortunately, it is very reluctant to produce berries, so it is a bit of a dead end from a breeding perspective. It was selected over three generations, first crossing a chromosome doubled 8x(4EBN) accession of S. acaule with Katahdin, then crossing the progeny of that cross with Ontario, and finally crossing the progeny of that cross with a tuberosum breeding line. Alaska Frostless is reported to be pentaploid, although the breeding history leads me to believe that a near-pentaploid aneuploid is more likely. The expected result from the first generation would be a 6x(4EBN) hybrid with 4 copies of the ACL genome and 2 copies of the TUB genome. The expected result from the second generation cross would be a 5x(4EBN) hybrid with 2 copies of the ACL genome and 3 copies of the TUB genome. The final cross would be expected to produce a 4.5x(4EBN) hybrid with 2 copies of the ACL genome and ~2.5 copies of the TUB genome. A true pentaploid isn’t impossible, though, just unlikely. If it is an aneuploid, it is quite likely to be 3 or 3.5 EBN, which might explain its apparent sterility. I was able to obtain a few seeds from a single berry in 2019, but this is unfortunately not a variety that is promising for breeding. S. curtilobum varieties have a similar genetic composition and comparable frost resistance but much greater fertility.
There are five common methods for making crosses between Solanum acaule and tetraploid S. tuberosum: (1) Pollinating S. acaule with a diploid S. tuberosum and then chemically chromosome doubling the progeny; (2) Chemically doubling S. acaule and then pollinating with tetraploid S. tuberosum; (3) Pollinating S. acaule with a diploid, 2EBN wild species, chemically chromosome doubling the progeny, and then crossing to tetraploid S. tuberosum; (4) Directly pollinating S. acaule with 4x S. tuberosum pollen and hoping that S. acaule produces some unreduced ovules; (5) Artificially combining S. acaule and tetraploid S. tuberosum through protoplast fusion.
S. tuberosum can be crossed to S. acaule when S. acaule produces unreduced ovules. The unreduced ovule of S. acaule is 4x and 4 EBN and the normal pollen of S. tuberosum is 2x and 4 EBN. This results in a hexaploid (6x), 4EBN hybrid. S. acaule cannot be crossed to S. tuberosum with much probability of success because it rarely produces unreduced pollen. Camadro (1990) reported success in crossing 4x S. tuberosum to the hexaploid progeny, but no success in the reciprocal cross, even though the plants appeared to produce viable pollen.
In at least one case mentioned by Ochoa (2004), crossing the diploid S. tuberosum (as S. goniocalyx) cultivar Tumbay to S. acaule resulted in self-fertile tetraploid progeny. There might be unrealized potential in crosses between diploid andigena varieties and S. acaule, which can be easily performed by amateurs.
The most commonly used method for crossing with S. tuberosum is to cross S. acaule x 4x S. tuberosum, chemically double the triploid hybrid to 6x/4EBN, and then cross again to 4x S. tuberosum, yielding mostly tetraploids. Because this method involves at least two crosses with S. tuberosum, the progeny are only 1/4 S. acaule. In practice, it often requires more than one backcross to S. tuberosum to get a true tetraploid, further reducing to the contribution of S. acaule to 1/8 or 1/16.
S. acaule can be crossed in either direction with diploid S. tuberosum. The gametes of S. acaule are 2x and 2EBN and the gametes of diploid S. tuberosum are 1x and 2EBN, resulting in hybrids that are triploid (3x) and 2EBN. The triploid progeny can be chemically doubled, yielding 4EBN hexaploids than can be crossed with 4x S. tuberosum.
Although out of the reach of most amateurs, it is also possible to hybridize S. acaule with S. tuberosum via embryo rescue and protoplast fusion. Direct 4x S. tuberosum x S. acaule crosses can be made with the assistance of embryo rescue. You can find the details in Iwanaga (1991).
| Female | Male | Berry Set |
Seed Set | Germ | Ploidy | Source |
|---|---|---|---|---|---|---|
| S. acaule | S. tuberosum | Low | Very Low | Low | Hexaploid | Camadro 1990 |
| S. acaule | S. tuberosum | Moderate | Moderate | Jackson 1999 | ||
| S. acaule | S. tuberosum 2x | Moderate | High | Tetraploid | Ochoa 2004 | |
| S. acaule | S. tuberosum 4x | Low | Low | Ochoa 2004 | ||
| S. tuberosum | S. acaule | None | Camadro 1990 | |||
| S. tuberosum | S. acaule | Minimal | None | Jackson 1999 | ||
| S. tuberosum 4x | S. acaule | Low | Moderate | Ochoa 2004 |
Crosses with other species
Solanum acaule crosses fairly easily with South American diploid species with an EBN of 2.
Watanabe (1991) found that 1.3% (only one plant) of varieties of this species produced 2n pollen and Jackson (1999) found 5-20%, which would be effectively octoploid and 4EBN.
According to Dionne (1963), S. acaule has been used as a bridge species to bring traits from 1EBN Central American diploid species into compatibility with S. tuberosum. The 1EBN diploids are used to pollinate S. acaule and the resulting hybrids can then be used to pollinate tetraploid S. tuberosum. The hybrids were triploid and sterile, but became fertile after chromosome doubling with colchicine.
| Female | Male | Berry Set |
Seed Set | Germ | Ploidy | Source |
|---|---|---|---|---|---|---|
| S. acaule | S. albicans | High | Low | Ochoa 2004 | ||
| S. acaule | S. berthaultii | High | High | Ochoa 1990 | ||
| S. acaule | S. boliviense (as S. megistacrolobum) | Some | Some | Ochoa 1990 | ||
| S. acaule | S. boliviense (as S. megistacrolobum) | High | None | Ochoa 2004 | ||
| S. acaule | S. boliviense (as S. megistacrolobum) | Low | Low | Low | Triploid | Okada 1982 |
| S. acaule | S. brevicaule 2x (as S. gourlayi) | High | High | High | Triploid | Camadro 1995 |
| S. acaule | S. brevicaule 4x (as S. gourlayi) | Low | Very Low | Very Low | Tetraploid | Camadro 1995 |
| S. acaule | S. bulbocastanum | Low | Very Low | Low | Hermsen 1966 | |
| S. acaule | S. candolleanum (as S. bukasovii) | High | Moderate | Triploid | Ochoa 1990, Ochoa 2004 | |
| S. acaule | S. chomatophilum | High | Moderate | Ochoa 2004 | ||
| S. acaule | S. commersonii | Low | Very Low | Triploid | Camadro 1995 | |
| S. acaule | S. raphanifolium | High | High | Ochoa 2004 | ||
| S. acaule | S. sogarandinum | High | Moderate | Ochoa 2004 | ||
| S. albicans | S. acaule | High | Low | Ochoa 2004 | ||
| S. berthaultii | S. acaule | Low | None | Ochoa 1990 | ||
| S. boliviense (as S. megistacrolobum) | S. acaule | High | None | Ochoa 2004 | ||
| S. brevicaule 2x (as S. gourlayi) | S. acaule | None | Camadro 1995 | |||
| S. brevicaule 4x (as S. gourlayi) | S. acaule | None | Camadro 1995 | |||
| S. candolleanum (as S. bukasovii) | S. acaule | High | Moderate | Ochoa 2004 | ||
| S. chomatophilum | S. acaule | High | Moderate | Ochoa 2004 | ||
| S. sogarandinum | S. acaule | High | Moderate | Ochoa 2004 |
References
Solanum acaule at Solanaceae Source

































